IRWD Q4 Earnings and Revenues Fall Shy of Estimates, Stock Tanks
Ironwood Pharmaceuticals IRWD reported an adjusted loss of 1 cent per share for the fourth quarter of 2025, against the Zacks Consensus Estimate of earnings of 2 cents. The company had reported adjusted earnings of 1 cent per share in the year-ago quarter.
Total revenues in the fourth quarter were $47.7 million, which also missed the Zacks Consensus Estimate of $53 million. Revenues decreased around 47.3% year over year.
Shares of Ironwood were down 11.4% yesterday, probably due to weaker-than-expected results.
In the past six months, shares of Ironwood have rallied 166.7% against the industry’s decline of 1.1%.
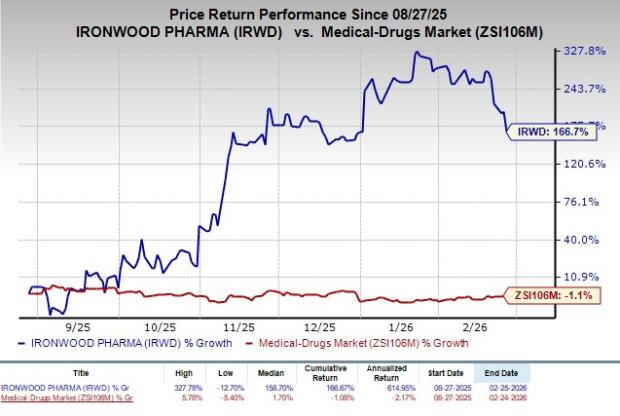
Image Source: Zacks Investment Research
IRWD's Q4 Earnings in Details
As reported by its partner AbbVie ABBV, Ironwood’s sole marketed product, Linzess (linaclotide), generated net sales of $163.2 million in the United States, down 27% year over year, due to rebate timing issues and higher pricing pressure from the Medicare Part D redesign.
Total prescription demand for Linzess increased 13% year over year during the fourth quarter.
IRWD and ABBV equally share Linzess’ brand collaboration profits and losses.
Ironwood’s share of net profit from the sales of Linzess in the United States (included in collaborative revenues) totaled $45.2 million, reflecting a decline of 49% year over year.
The company has agreements with two partners, Astellas Pharma and AstraZeneca AZN, related to the development and commercialization of Linzess in Japan and China, respectively.
Astellas and AstraZeneca have exclusive rights to develop and market the drug in their respective territories. Both companies are liable to pay royalties to Ironwood on net Linzess revenues earned in their regions.
Ironwood's royalties and other revenues were $2.5 million in the fourth quarter. In the year-ago quarter, the company recorded $2.1 million in royalties and other revenues.
Total cost and expenses (including research and development expenses, selling, general and administrative expenses and restructuring expenses) in the fourth quarter were $40.9 million, down 30.8% from the year-ago quarter.
Ironwood recorded adjusted EBITDA of $10.9 million in the fourth quarter, reflecting a 70.7% decrease year over year.
As of Dec. 31, 2025, Ironwood had cash and cash equivalents worth $215.5 million compared with $140.4 million as of Sept. 30, 2025.
IRWD's Full-Year Results
For 2025, Ironwood generated revenues of $296.2 million, decreasing around 15.7% on a year-over-year basis.
For the same period, the company reported adjusted earnings of 25 cents per share, up from 4 cents reported in the year-ago period.
IRWD's 2026 Guidance
Ironwood reiterated its full-year 2026 revenue guidance, which it had provided last month.
The company continues to expect total revenues of $450 million to $475 million in 2026.
U.S. sales of Linzess (to be recorded by AbbVie) are expected to be in the range of $1.13-$1.18 billion in 2026.
The company expects to deliver an adjusted EBITDA of more than $300 million in 2026, indicating effective cost management.
Ironwood expects a significant improvement in Linzess’ sales in 2026 and subsequently its share of net profit from the sales of this partnered drug in the United States.
Also, effective Jan. 1, 2026, Linzess’ list price was reduced to help maintain patient access. Despite the price cut, management expects Linzess’ net sales to increase year over year in 2026, as the lower list price will reduce certain mandatory government rebates. Fewer rebate payments should translate into higher net revenues in 2026.
IRWD's Recent Pipeline Updates
Ironwood is developing its next-generation GLP-2 analog, apraglutide, for treating patients with short bowel syndrome (“SBS”) with intestinal failure (“IF”) who are dependent on parenteral support (“PS”).
Ironwood recently met with the FDA and aligned on key elements of a confirmatory phase III study design (to be called STARS-2), which will evaluate apraglutide in patients with SBS-IF. The primary endpoint of the STARS-2 study will check the relative change from baseline in actual weekly PS volume at week 24 in the given patient population.
Per management, initiation of clinical sites for the STARS-2 study is expected to begin in the second quarter of 2026 with a new drug application submission targeted before year-end 2029.
Ironwood acquired the rights to develop and commercialize apraglutide following the acquisition of VectivBio in June 2023.
Ironwood Pharmaceuticals, Inc. Price, Consensus and EPS Surprise
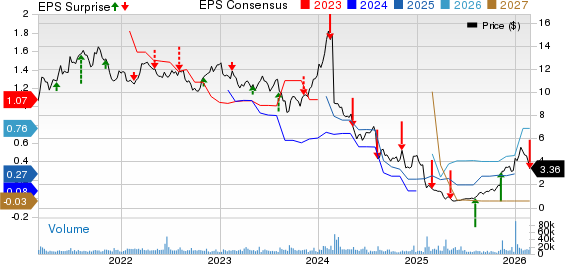
Ironwood Pharmaceuticals, Inc. price-consensus-eps-surprise-chart | Ironwood Pharmaceuticals, Inc. Quote
IRWD’s Zacks Rank & Stock to Consider
Ironwood currently carries a Zacks Rank #3 (Hold).
A better-ranked stock in the biotech sector is Castle Biosciences CSTL, currently sporting a Zacks Rank #1 (Strong Buy). You can see the complete list of today’s Zacks #1 Rank stocks here.
Over the past 60 days, Castle Biosciences’ 2026 loss per share estimates have narrowed from $1.06 to 96 cents. CSTL’s shares have rallied 44% over the past six months.
Castle Biosciences’ earnings beat estimates in three of the trailing four quarters and missed in the remaining one, with the average surprise being 66.11%.
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
NexPoint (NREF) Matches Q4 Earnings Expectations
TriMas (TRS) Falls Short of Q4 Earnings Expectations
D-Wave Quantum Inc. (QBTS) Posts Fourth Quarter Loss, Falls Short of Revenue Expectations
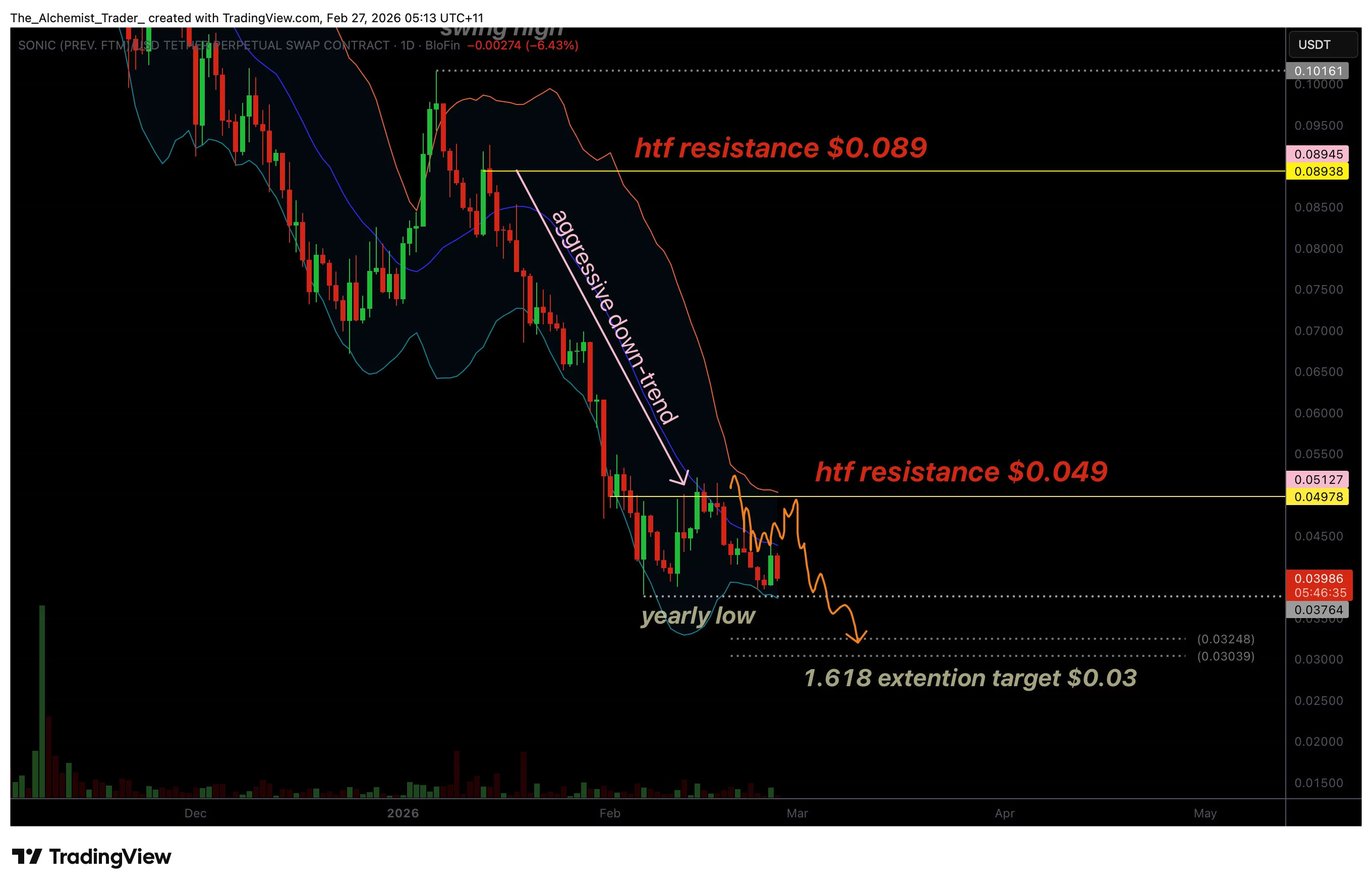
Sonic price eyes new yearly lows as aggressive downtrend continues