REGN & SNY's Dupixent Receives CHMP Approval for Pediatric Treatment of Urticaria
Dupixent Receives Positive CHMP Recommendation for Pediatric Urticaria
Regeneron Pharmaceuticals and Sanofi have announced that the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has issued a favorable opinion regarding the expanded use of Dupixent. This recommendation supports the drug’s use in children aged 2 to 11 with moderate-to-severe chronic spontaneous urticaria (CSU) who have not responded adequately to histamine-1 antihistamines and have not previously received anti-immunoglobulin E therapy. The European Commission is expected to make a final decision in the near future.
Dupixent has already been approved in the European Union for CSU treatment in adults and adolescents aged 12 and older since November 2025.
Chronic spontaneous urticaria is a skin disorder characterized by type II inflammation, resulting in sudden outbreaks of hives and swelling. These symptoms are often difficult to manage with antihistamines alone.
In addition to CSU, Dupixent is authorized in the United States, European Union, Japan, and other regions for a variety of conditions, including asthma, atopic dermatitis, chronic rhinosinusitis with nasal polyps, chronic obstructive pulmonary disease, eosinophilic esophagitis, prurigo nodularis, bullous pemphigoid, and allergic fungal rhinosinusitis across multiple age groups.
Currently, Dupixent is not approved in the United States for CSU in children. The FDA is reviewing a supplemental biologics license application from Sanofi and Regeneron for use in patients aged 2 to 11, with a decision expected by April 2026.
Over the past year, Regeneron’s stock has increased by 14.1%, while the broader medical and genetics industry has seen a 20.1% rise.
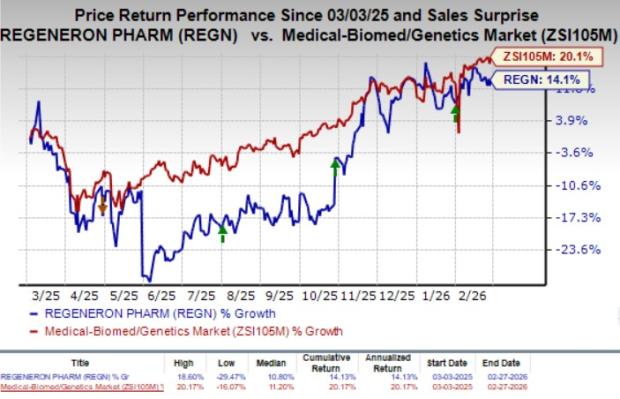
Image Source: Zacks Investment Research
CHMP Approval Supported by Phase III Clinical Data
The CHMP’s positive recommendation for Dupixent in pediatric CSU is based on results from two advanced clinical trials: Study A and Study C, part of the phase III LIBERTY-CUPID program and the CUPIDKids study.
Findings from the LIBERTY-CUPID program demonstrated that Dupixent significantly reduced itching and hives compared to placebo after 24 weeks. The treatment also led to a higher proportion of patients achieving well-controlled disease and complete response versus placebo.
Global Collaboration and Revenue Highlights
Regeneron and Sanofi maintain a worldwide partnership for the discovery, development, and commercialization of Dupixent, Kevzara, and itepekimab. Under this agreement, Sanofi records global net sales for Dupixent and Kevzara, while Regeneron reports its share of profits or losses related to these sales.
In 2025, Regeneron’s collaboration revenue from Sanofi reached $5.9 billion, primarily driven by Dupixent, marking a 30% increase from the previous year.
Sanofi reported global Dupixent sales of $17.8 billion (€15.7 billion) in 2025, reflecting a 26% year-over-year growth.
Regeneron’s Zacks Rank and Biotech Stock Recommendations
Regeneron Pharmaceuticals currently holds a Zacks Rank #3 (Hold).
Among leading biotech stocks, Castle Biosciences is rated Zacks Rank #1 (Strong Buy), and Assertio Holdings is rated Zacks Rank #2 (Buy).
- Castle Biosciences’ projected loss per share for 2026 has improved from $1.06 to $0.96 over the past 60 days. CSTL shares have climbed 39% in the last year.
- Castle Biosciences exceeded earnings expectations in three of the past four quarters, with an average positive surprise of 34.69%.
- Assertio’s estimated loss per share for 2026 has narrowed from $0.30 to $0.28 in the last 60 days. ASRT shares are up 1% year-over-year.
- Assertio surpassed earnings estimates in one of the previous four quarters, but missed in the other three, with an average negative surprise of 35.21%.
Five Stocks Poised for Significant Growth
Zacks experts have selected five stocks expected to potentially double in value in the coming months:
- Stock #1: A dynamic company demonstrating strong growth and resilience
- Stock #2: Showing bullish indicators and ripe for buying on dips
- Stock #3: Considered one of the most attractive investments currently available
- Stock #4: Leading a rapidly expanding industry
- Stock #5: A modern omni-channel platform ready for expansion
Many of these stocks are not widely recognized on Wall Street, offering early investment opportunities. Previous recommendations have achieved gains of +171%, +209%, and +232%.
Free Stock Analysis Reports
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
Developer Says $100 XRP Price Is Not an Insane Prediction. Here’s Why
Decoding Bitcoin’s capital rotation – $5B retail exits as whales take control
FirstEnergy Secures $950M Transmission Project: A Strategic Move Toward Grid Upgrades
Apple unveils the more affordable iPhone 17e in an effort to increase iPhone sales

