Mapping the S-Curve: INOVIO's Partnership in GBM as a Strategic Move Toward Advancing DNA Immunotherapy Platforms
Glioblastoma: A High-Risk Frontier in Brain Cancer Treatment
Glioblastoma (GBM) stands as the most formidable form of brain cancer, notorious for its aggressive progression. Patients face a median survival of about 15 months, and the odds of surviving five years are less than three percent. This grim outlook highlights the urgent need for new therapies and fuels ongoing research efforts. INOVIO has partnered with Regeneron to develop INO-5401, aiming to create a new foundation for treating these challenging tumors. However, this initiative is still in its early, uncertain stages, where the risks are high and success is far from assured.
Current clinical results illustrate the uphill battle. INO-5401 is undergoing Phase II trials for GBM, but only 23% of drugs in this category advance successfully at this stage. This rate is lower than the average for similar treatments, indicating that INO-5401 faces a particularly tough road. The drug remains in the experimental phase, where setbacks are common and proven effectiveness has yet to be demonstrated. INOVIO is essentially wagering on a transformative breakthrough before the technology has proven itself at scale.
Investor confidence is reflected in INOVIO’s stock performance, which recently dipped to $1.68 per share. This price signals skepticism about both the clinical prospects and financial sustainability of the company’s pipeline. Such volatility is typical for firms operating at the cutting edge of innovation, where the likelihood of failure is high. The collaboration represents a bold gamble that DNA-based immunotherapy could become a cornerstone of future cancer treatments, but for now, the market is waiting for tangible results.
Innovative Approaches: Laying the Groundwork for Next-Gen Immunotherapy
INOVIO’s alliance with Akeso is a calculated move to advance the infrastructure of immunotherapy. The strategy involves combining INOVIO’s DNA technology with a pioneering checkpoint inhibitor to more effectively target cancer’s ability to evade the immune system. The clinical trial will pair INO-5412—a DNA vaccine designed to target tumor-specific antigens—with Akeso’s cadonilimab, a bispecific antibody that blocks both PD-1 and CTLA-4 checkpoints. This dual approach is intended to stimulate a stronger T-cell response, transforming GBM’s typically “cold” tumor environment into one that is more susceptible to immune attack.
The INSIGhT adaptive platform trial, which will evaluate this combination, is structured to test multiple therapies efficiently by using a shared control group. This design can significantly speed up development and lower costs, which is crucial when pioneering new treatment models. The scientific rationale is compelling: DNA immunotherapy primes the immune system, while checkpoint inhibitors remove inhibitory signals. Together, they may overcome the limitations of each approach alone. Previous Phase II results with INO-5401 and a PD-1 inhibitor showed promising immune responses and potential survival benefits, providing a solid basis for this next step.
However, a major challenge remains. While cadonilimab has demonstrated effectiveness in other cancers and has been approved in China for gastric and cervical cancers, its impact on GBM—a cancer with a highly suppressive immune environment—remains untested. This trial will serve as a crucial test of whether the combination can succeed in one of the most difficult cancer types. A positive outcome could establish a new standard for combination immunotherapies, while failure would underscore the biological complexities of GBM and the risks of innovative approaches. For now, INOVIO is leveraging a high-efficiency platform to rigorously evaluate this promising, yet unproven, technology.
Financial Challenges and Execution Risks
The financial outlook for INOVIO’s GBM program is closely tied to its ability to secure funding and achieve meaningful clinical milestones. Previous Phase II data combining INO-5401 with cemiplimab showed a median overall survival of 32.5 months in MGMT-methylated patients, a notable improvement over historical outcomes. This sets a high bar for the new combination with cadonilimab, which must deliver a statistically significant survival benefit beyond this benchmark to justify further investment and adoption.
This scenario introduces significant execution risk. The Akeso partnership is a resource-intensive endeavor, as Phase II trials demand considerable funding for patient enrollment, monitoring, and data analysis. INOVIO’s ability to continue the trial depends on its financial reserves and future fundraising efforts. Without sufficient capital, the study could face delays or reductions in scope, jeopardizing the entire infrastructure-building strategy. The market’s wariness, as seen in the depressed stock price, reflects these concerns.
Competition adds another layer of complexity. Akeso’s ivonescimab, for example, showed only modest improvements over Keytruda and failed to achieve a statistically significant survival benefit in interim analysis. This highlights the high standards required to prove a new therapy is superior to established treatments. For INOVIO, the new combination must not only work but also clearly outperform its own previous results. Falling short would reinforce investor caution and cast doubt on the near-term promise of DNA immunotherapy.
Ultimately, the financial sustainability of this high-risk venture depends on two factors: securing enough funding to advance the trial and generating transformative clinical data. Until both are achieved, the collaboration remains a bold but unproven effort, exposed to the same pitfalls that have challenged many biotech innovators.
Key Milestones and Risks Ahead
The future of this collaboration is shaped by several pivotal events and uncertainties. The most immediate catalyst is the interim results from the INSIGhT trial, which will offer early insights into the safety and effectiveness of the therapy. This adaptive trial is designed for rapid evaluation, and its initial findings will be crucial in determining whether the infrastructure approach is viable. A positive outcome could accelerate development, while disappointing results may halt further investment in this combination.
Another important factor is Akeso’s progress with its other bispecific antibody programs. The company recently received a fifth Breakthrough Therapy Designation for ivonescimab in biliary tract cancer, reflecting its commitment to advancing multiple assets. This momentum could strengthen Akeso’s support for the cadonilimab program, but it also means resources may be divided among competing priorities. Any setbacks in Akeso’s broader pipeline could indirectly affect the pace and focus of the GBM collaboration.
The greatest challenge remains clinical validation. The new therapy must show a clear survival advantage over the current standard, which includes INOVIO’s own prior data of a 32.5-month median survival for certain patients. With only a 23% success rate for GBM drugs advancing through clinical phases, the bar is set extremely high. Failure to achieve a significant benefit could end further development and raise doubts about the viability of DNA immunotherapy in the near term.
Patient enrollment for the combination therapy trial is expected to start in the second half of 2026, setting a clear timeline for the next major data release. For investors, this represents a classic high-stakes scenario: the collaboration is laying the tracks for a new approach to cancer treatment, but its future depends on whether the first results can deliver. The coming months will reveal whether this is the beginning of a new era in cancer care or a costly detour.
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
Investors Are Actively Looking Into NextEra Energy, Inc. (NEE): Key Information You Should Be Aware Of
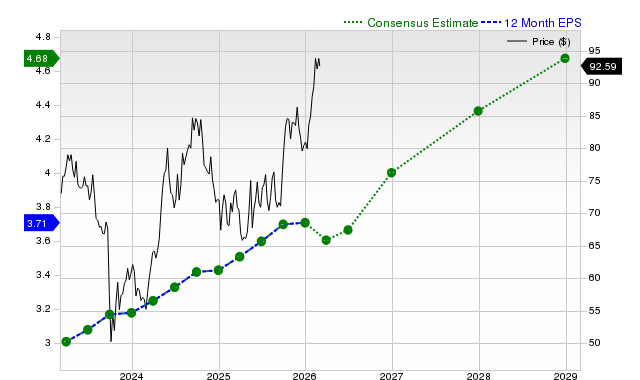
Can Quanta Leverage Data Center Demand Patterns to Boost Profit Margins?
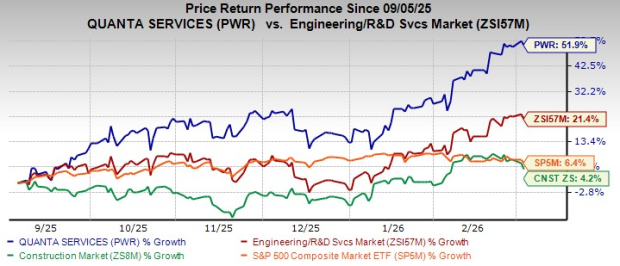
RedStone launches Stellar price feeds after $10M exploit highlights oracle risks
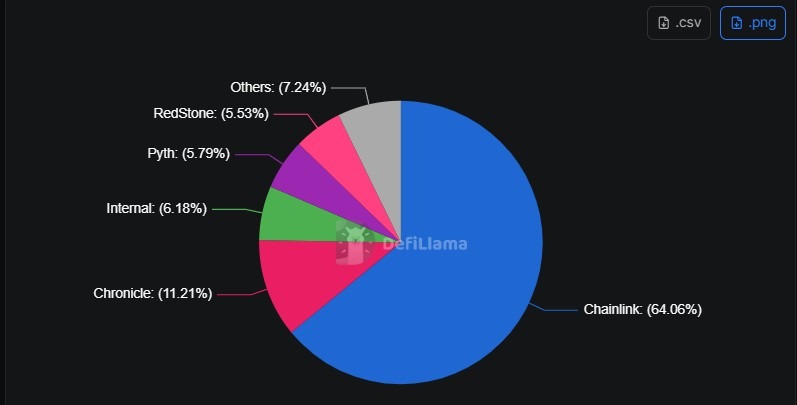
RDN Shares Priced Below Industry Average at 0.98X: Is Now the Right Time to Invest?