BMY Wins FDA Nod to Expand Sotyktu Label for Psoriatic Arthritis
Bristol Myers Squibb BMY has announced that the FDA approved a label expansion of psoriasis drug, Sotyktu (deucravacitinib), an oral, selective tyrosine kinase 2 (TYK2) inhibitor.
The regulatory body approved the drug for the treatment of adults with active psoriatic arthritis (PsA).
Per BMY, the latest approval makes Sotyktu the first and only TYK2 inhibitor to be approved for this indication.
Shares of the company have lost 4.5% in the past year against the industry’s growth of 12.9%.
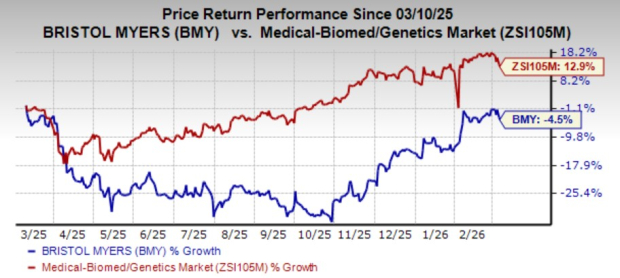
Image Source: Zacks Investment Research
More on BMY’s Sotyktu
The FDA approval was based on positive results from the POETYK PsA-1 and POETYK PsA-2 studies, which evaluated the efficacy and safety of Sotyktu 6 mg once daily in adults with active psoriatic arthritis.
In both studies, Sotyktu achieved the primary endpoint by significantly improving disease activity compared with placebo at week 16.
In the PsA-1 study, 54% of patients receiving Sotyktu achieved an American College of Rheumatology (ACR) 20 (the primary endpoint) response compared with 34% for placebo. Similarly, the PsA-2 trial showed a 54% response rate versus 39% for placebo.
Higher response thresholds also showed encouraging results. In PsA-1, ACR50 and ACR70 response rates were 24% and 12%, respectively, compared with 14% and 5% for placebo. Comparable improvements were observed in PsA-2.
Patients also demonstrated meaningful improvement in minimal disease activity and physical quality-of-life scores.
We remind investors that the FDA approved Sotyktu in 2022 for the treatment of adults with moderate-to-severe plaque psoriasis who are candidates for systemic therapy or phototherapy. Sotyktu has five years of clinical efficacy and safety data in patients with moderate-to-severe plaque psoriasis.
The drug raked in sales of $291 million in 2025, up 19% from 2024. The label expansion of the drug will boost sales.
BMY Focuses on Diversifying Its Portfolio
Bristol Myers delivered a resilient performance in 2025, supported by strong contributions from key growth drivers, such as Opdivo, Opdualag, Reblozyl, Breyanzi and Camzyos. These products helped stabilize the company’s revenue base despite ongoing generic erosion across its legacy portfolio.
Looking ahead, potential approvals of new drugs and label expansions for existing drugs should further diversify revenue streams.
In addition, upcoming pipeline readouts represent meaningful near-term catalysts that could strengthen the long-term growth outlook of BMY’s growth portfolio.
Last year, Bristol Myers collaborated with Bain Capital to create a new independent biopharmaceutical company focused on developing therapies for autoimmune diseases that address significant unmet needs of patients.
The new firm started with five immunology assets licensed from BMY and a $300 million investment led by Bain Capital.
The pipeline includes three clinical-stage and two phase I-ready programs targeting key immune pathways. Leading assets include afimetoran, an oral TLR7/8 inhibitor currently in phase II trials for systemic lupus erythematosus, and BMS-986322, an oral TYK2 inhibitor that showed positive phase II results in plaque psoriasis.
BMY’s Sotyktu faces stiff competition from Amgen’s AMGN Otezla in the psoriasis space. Notably, Amgen acquired global commercial rights to Otezla from erstwhile Celgene (now part of Bristol-Myers).
Amgen recorded Otezla sales of $2.26 billion in 2025.
BMY’s Zacks Rank & Key Picks
BMY currently carries a Zacks Rank #3 (Hold). Some better-ranked stocks in the drug/biotech sector are Catalyst Pharmaceuticals CPRX and ANI Pharmaceuticals ANIP. While Catalyst Pharmaceuticals currently sports a Zacks Rank #1 (Strong Buy), ANI Pharmaceuticals holds a Zacks Rank #2 (Buy). You can see the complete list of today’s Zacks #1 Rank stocks here.
Over the past 60 days, estimates for Catalyst Pharmaceuticals’ 2026 earnings per share have increased from $2.53 to $2.82, and the same for 2027 have grown from $2.85 to $3.20. CPRX’s shares have risen 17% in the past year.
Catalyst Pharmaceuticals’ earnings beat estimates in each of the trailing four quarters, with the average surprise being 35.19%.
Over the past 60 days, estimates for ANI Pharmaceuticals’ 2026 earnings per share have increased from $8.20 to $9.00, while the same for 2027 have risen from $9.25 to $10.10. ANIP’s shares have gained 25.9% in the past year.
ANI Pharmaceuticals’ earnings beat estimates in each of the trailing four quarters, with the average surprise being 22.21%.
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
Why XRP's Long-Term Vision Lies In The Internet Of Value Stack

BC-Gold-Silver
Crisis por rotura de gasoducto clave pone a prueba resiliencia de la industria en Perú
Savings Accounts Vs. Dividend ETFs: How A $10,000 Investment Could Grow Over 10 Years
