AEON Biopharma Appoints John Bencich as Chief Financial Officer
Veteran biotechnology leader with extensive capital markets experience joins AEON as Company advances lead asset through key regulatory milestones
IRVINE, Calif., March 09, 2026 (GLOBE NEWSWIRE) -- AEON Biopharma, Inc. (“AEON” or the “Company”) (NYSE American: AEON), a biopharmaceutical company advancing ABP-450 (prabotulinumtoxinA) as a biosimilar to BOTOX
“John’s appointment represents an important step forward for AEON as we enter a period of accelerated execution,” said Rob Bancroft, President and Chief Executive Officer of AEON. “His deep experience leading finance organizations at publicly traded biotechnology companies, combined with his capital markets expertise, will strengthen our financial strategy, deepen institutional engagement, and support disciplined capital formation as we advance our regulatory pathway. We believe John’s leadership will be instrumental in positioning AEON for long-term value creation.”
“AEON is executing a focused and differentiated biosimilar strategy within a large and attractive therapeutic market,” said Mr. Bencich. “I see significant opportunity to strengthen the Company’s financial foundation and support the next stage of growth to help fully realize its potential. I am excited to join the team at such a defining moment.”
Mr. Bencich joins AEON with more than 25 years of leadership experience spanning corporate strategy, capital market transactions, and business development across emerging growth and publicly traded companies. Most recently, he served as Chief Executive Officer of Achieve Life Sciences, where he led the company’s capitalization and helped drive significant market capitalization growth. During his tenure, he also oversaw key clinical and regulatory milestones, strengthened the company’s intellectual property portfolio, and advanced commercial and business development initiatives. Earlier in his career, he held Chief Financial Officer positions at OncoGenex Pharmaceuticals, Integrated Diagnostics, Allozyne, and Trubion Pharmaceuticals, where he supported multiple financings, strategic transactions, and public-company readiness initiatives. He holds a Bachelor of Accountancy from the University of San Diego and an MBA from Seattle University.
About the U.S. Biosimilar Pathway
Under the FDA’s 351(k) biosimilar pathway, developers must demonstrate that a proposed product is highly similar to an approved reference biologic, with no clinically meaningful differences in safety, purity, or potency. Analytical similarity represents the scientific foundation of this process, integrated with clinical and regulatory strategy to form a totality-of-evidence assessment. FDA engagement focuses on determining the scope of data necessary to address residual uncertainty, which may include analytical, nonclinical, or clinical components as appropriate.
About AEON Biopharma
AEON Biopharma is a biopharmaceutical company advancing a strategy to achieve accelerated and full-label access to the U.S. therapeutic neurotoxin market through biosimilarity to BOTOX
Forward-Looking Statements
Certain statements in this press release may be considered forward-looking statements. Forward-looking statements generally relate to future events or AEON’s future financial or operating performance and may include statements regarding the Company’s regulatory plans, development strategy for ABP-450, anticipated capital formation activities, growth opportunities, and the expected contributions of Mr. Bencich to the Company’s strategy and execution.
These statements are based on current expectations and assumptions and are subject to risks and uncertainties that could cause actual results to differ materially from those expressed or implied by such forward-looking statements. Such risks and uncertainties include, among others, regulatory developments and outcomes, the Company’s ability to obtain necessary approvals, the availability of capital on acceptable terms, market conditions, competitive developments, and other risks described in the Company’s filings with the Securities and Exchange Commission.
Forward-looking statements speak only as of the date of this press release, and AEON undertakes no obligation to update such statements except as required by law.
Contacts
Source: AEON Biopharma
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
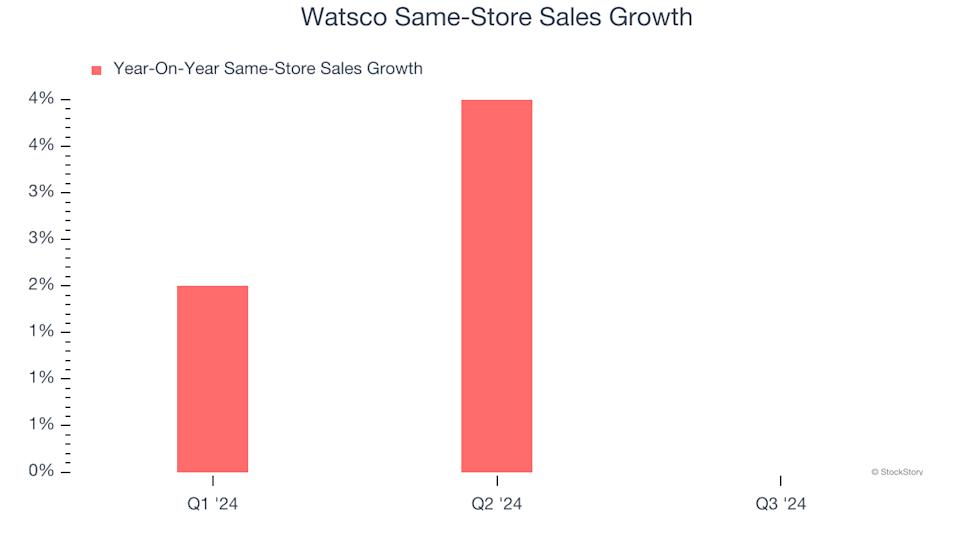
3 Reasons to Steer Clear of WSO and One Alternative Stock Worth Buying
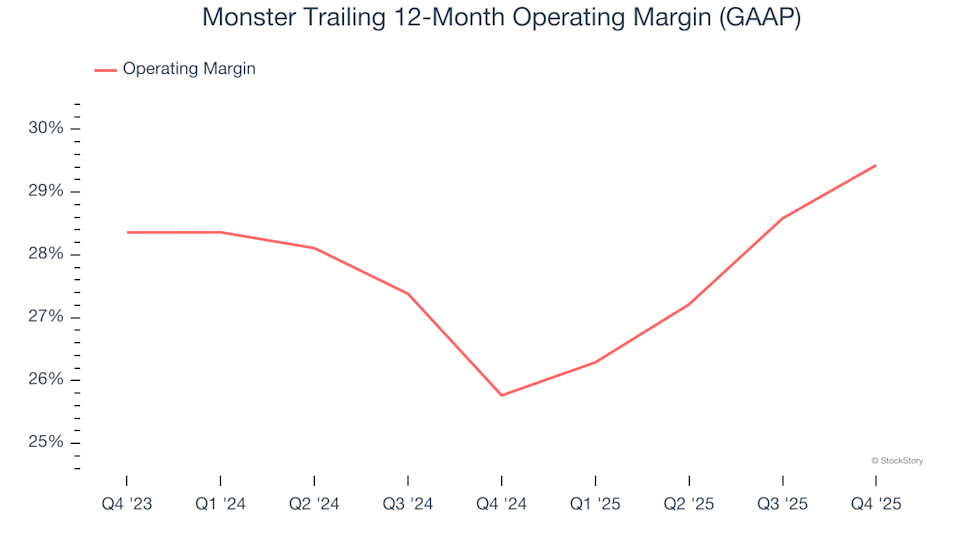
3 Key Factors That Make Investors Favor Monster (MNST)
Western Digital Corporation (WDC) Sets Sights on 50%+ Gross Margin and EPS $20+

Western Digital Corporation (WDC) Sets Sights on 50%+ Gross Margin and EPS $20+


