Sanofi’s Venglustat Receives FDA Breakthrough Status—Market Could Be Undervaluing Its Long-Term Potential
FDA Breakthrough Therapy Status: What It Means for Venglustat
Venglustat recently received Breakthrough Therapy designation from the FDA, a move that highlights the drug’s promising clinical results but signals the start of a lengthy development process rather than immediate financial gains.
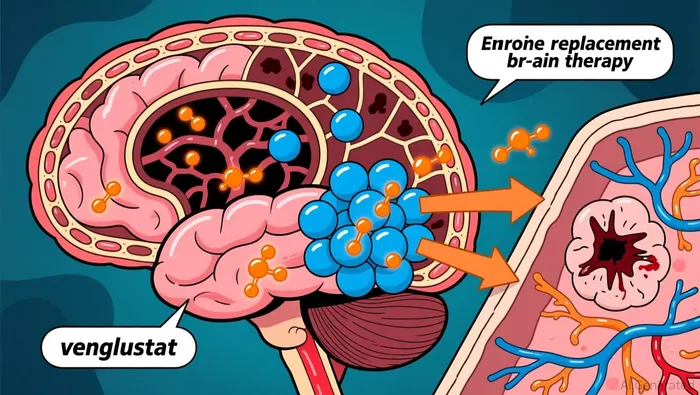
This designation was granted based on Phase 3 LEAP2MONO trial data, which demonstrated that venglustat outperformed enzyme replacement therapy (ERT) in treating neurological symptoms. The primary outcome was a statistically significant improvement in a comprehensive neurological assessment, with a p-value of 0.007. This result suggests venglustat is effective where current therapies fall short.
The focus is on chronic neuronopathic Gaucher disease (Type 3), a rare disorder with no approved therapies for neurological complications. While ERT can address systemic symptoms such as enlarged spleen and liver, it cannot penetrate the blood-brain barrier. Venglustat, an oral drug specifically designed to cross into the brain, aims to directly address the root cause of neurological decline. The FDA’s decision underscores the urgent need for new treatments in this area.
At this stage, the Breakthrough Therapy status accelerates the development and review process, allowing for more frequent engagement with the FDA. However, it does not eliminate the need for comprehensive safety and efficacy data. Approval remains several years away, with Sanofi targeting global regulatory submissions in 2026. While the designation affirms the scientific promise, it does not alter the timeline for potential commercial returns.
Assessing the Current Risk and Reward for Sanofi’s Stock
Although the Breakthrough Therapy designation is a positive development, it does not speed up the overall timeline. Venglustat is still in Phase 3, and regulatory filings are not expected until 2026. As the drug is intended for a rare, chronic condition, the addressable market remains limited.
This scenario creates a typical tactical opportunity. Sanofi’s stock is currently trading at €76.89, down 25.4% over the past year. This sharp drop reflects investor caution regarding the long wait for approval and the inherent risks of late-stage drug development, even with encouraging data. The recent regulatory milestone may not yet be fully reflected in the share price.
From another perspective, the current valuation could represent an opportunity. The stock is about 22% below the midpoint of analyst target ranges (€98.49), and some valuation models suggest it is trading roughly 75.6% below estimated fair value. Despite the pipeline progress, the market has not rewarded the stock, as shown by a slight decline over the past month.
Ultimately, this is a classic risk-versus-reward scenario. While the drug’s potential is significant, so are the challenges and the long wait for returns. The stock’s substantial decline may indicate excessive market pessimism about the timeline and market size, leaving room for a potential re-rating if development stays on course. For investors focused on catalysts, the regulatory validation is key, and the opportunity lies in the disconnect between this progress and the current share price.
Upcoming Milestones and What to Watch
The near-term outlook depends on several critical events that could either reinforce or challenge the investment case. The next major milestone is the submission of a New Drug Application (NDA) in the US and other regions, which will follow the Phase 3 results. Achieving this step would confirm the regulatory pathway and could serve as a significant short-term catalyst for the stock.
Investors should also keep an eye on the ongoing Phase 3 CARAT trial of venglustat for Fabry disease. While the earlier PERIDOT study did not meet its primary endpoint, the CARAT study provides further insight into the drug’s potential for another rare lysosomal storage disorder. Positive results could expand venglustat’s therapeutic reach and strengthen its overall profile.

Sanofi (SNY) is currently trading at 45.130 on NASDAQ, with a recent increase of +0.360 (+0.80%).

Risks and Key Considerations
- High development costs for rare disease drugs
- Potential competition from other innovative therapies
- Recent stock underperformance, with shares down 25.4% over the past year
This recent decline highlights ongoing market skepticism about the timeline and commercial potential, making future clinical and regulatory successes even more important.
Actionable Watchpoints
- Timing and outcome of global regulatory submissions in 2026
- Updates from the CARAT study in Fabry disease
- Stock performance in response to these milestones, especially given its current discount to analyst targets
In summary, the investment case hinges on successful execution and meeting key milestones. These upcoming events will be crucial in determining whether the current undervaluation presents a compelling opportunity.
Disclaimer: The content of this article solely reflects the author's opinion and does not represent the platform in any capacity. This article is not intended to serve as a reference for making investment decisions.
You may also like
Franklin Templeton Reveals Why They Bought XRP
Arm Unveils AI Chip: A Key Test for Its $140 Billion Market Value

Trade Desk's Fee Audit Frenzy: Analyzing the Sell-Off Through Fund Movements

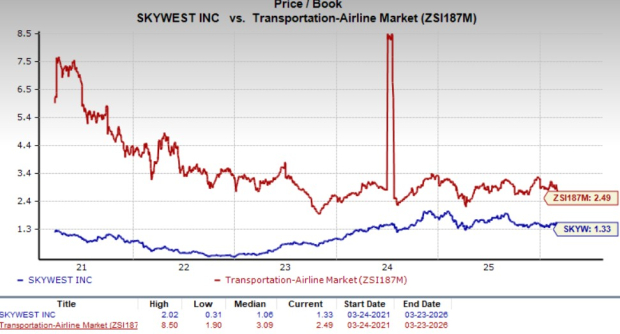
SkyWest's EPS Projections on the Rise: Is Now the Time for Investors to Purchase Shares?